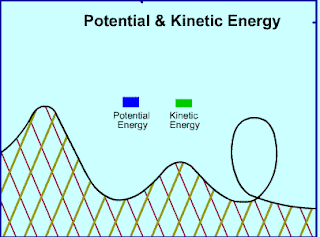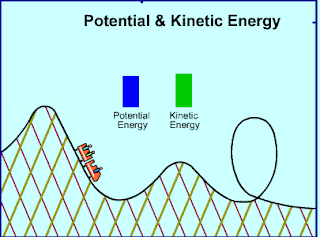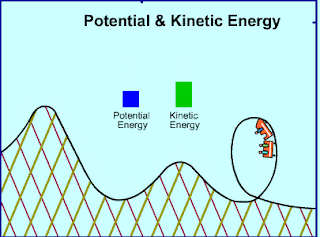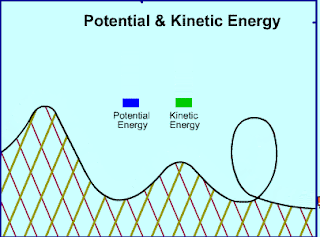
|
| Galaxy.... COOL! |
 |
| Another Galaxy! |
This story is important because it is a huge discovery for the astronomers. Because of this, I found it really importnant when i saw the article. I thouhgt it was cool when I saw that there was anew ghalaxy discovered, as well as it being the oldest and the farthest awaty from us that was found! I believe that they will use this disovery and continue on with it to try to find even more discoveries than then they already have. They have found so many discoveriws just dealing with space. What do you think they will discover next? I believe that they will either find some more things inside z8_GND_5296, or will find other galaxies and constelations.
Did you like my blog about Galaxy Discoveries?
Please tell me your feedback! I'd like to know how I did!
I would love to know what you thought of this!
Thanks for reading everyone! <3
Please tell me your feedback! I'd like to know how I did!
I would love to know what you thought of this!
Thanks for reading everyone! <3
xoxo Teemster! :D